New Publications!!
-
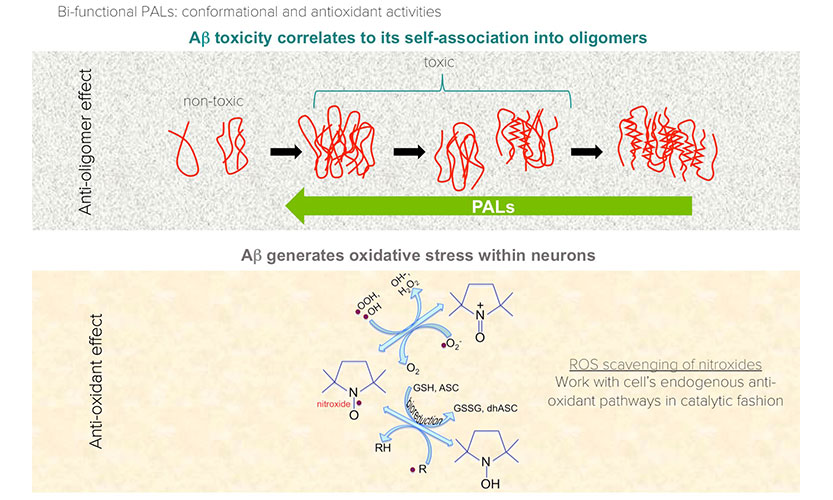
Novel Stilbene-Nitroxyl Hybrid Compounds Display Discrete Modulation of Amyloid Beta Toxicity and Structure
-
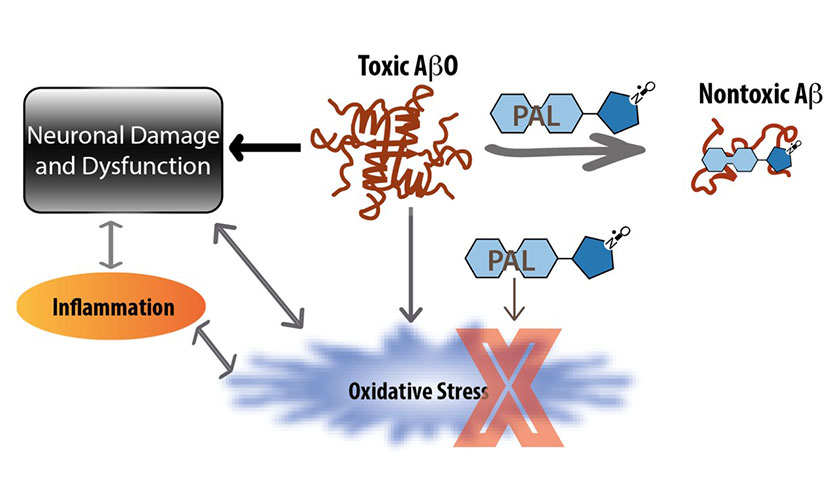
A Bifunctional Anti-Amyloid Blocks Oxidative Stress and the Accumulation of Intraneuronal Amyloid-Beta.
-
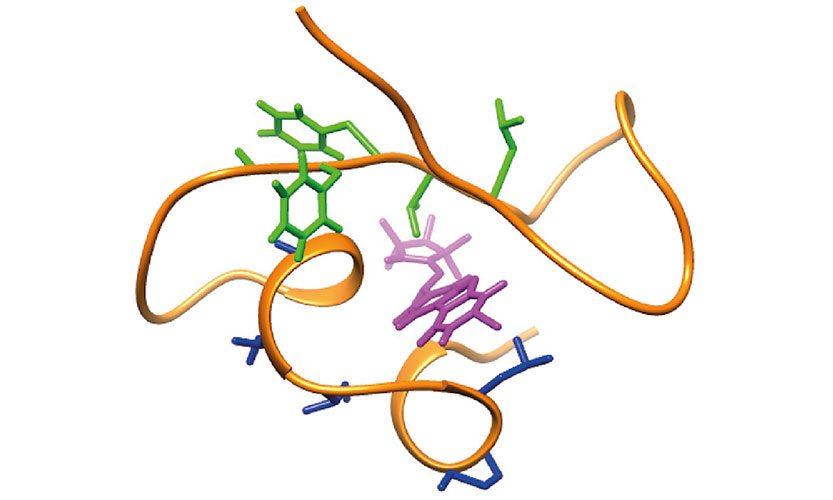
Completion of the Vimentin Rod Domain Structure Using Experimental Restraints: A New Tool for Exploring Intermediate Filament Assembly and Mutations.
At Voss Lab, we mainly focus on observing molecular switching in proteins, as well as protein folding and assembly. This work is carried out in large part through the use Electron Paramagnetic Resonance (EPR — a.k.a. ESR) spectroscopy of site-directed spin labels. We use this approach to address problems in several biological systems, including those related to cardiovascular and neurodegenerative diseases.
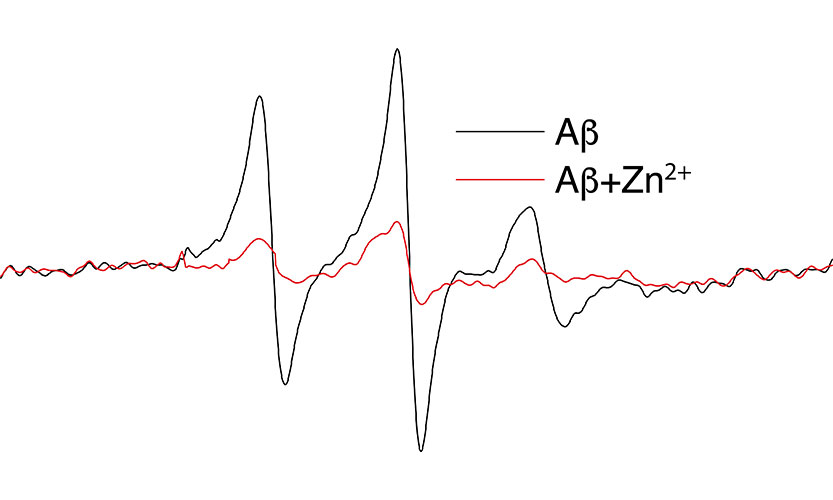
With spin labeling reagent, we can introduce a unique and stable species whose unpaired electrons produce a unique EPR signal. When the spin label is attached to a biological molecule the EPR signal (spectrum) reports on motions and structure within the molecule. This approach, known as site-directed spin-labeling (SDSL) represents one of the most powerful techniques currently available to investigate structure-function relationships in protein and DNA, the molecules whose activity provides the fundamental processes of life. In addition, EPR analysis can now be used in vivo, including imaging of structures visible only to EPR, providing a new Magnetic Resonance Imaging tool. The application of EPR technology is not limited to living systems. For example, because of its high sensitivity, EPR has recently been used to measure the quantity of energy used locally during a mechano-chemical milling process, semi-conductor doping, archeological dating, and food science. In summary, EPR spectroscopy is striving to bridge the gap between laboratory science and clinical medicine.