

Department of Physiology and Membrane Biology
Department of Anesthesiology and Pain Medicine
School of Medicine, University of California
1985-1993 |
Master of Science (M.S.) Degree in Physics, Department of Physics, Moscow State University, Moscow, Russia. |
1993-1998 |
Doctor of Philosophy (Ph.D.) Degree in Biochemistry and Molecular Biology, Department of Environmental and Biomolecular Systems, OGI School of Science & Engineering, Oregon Health Science University, Portland, Oregon. |
1999-2003 |
Senior Fellow in Computational Biology and Biophysics. Supervisors: Dr. David Baker, Professor, Department of Biochemistry, and Dr. William A. Catterall, Professor and Chairman, Department of Pharmacology, University of Washington, Seattle, Washington. |
2011-2015 |
Assistant Professor, Department of Physiology and Membrane Biology, University of California, Davis, CA |
2015-2020 |
Associate Professor, Department of Physiology and Membrane Biology, University of California, Davis, CA |
2020-present |
Professor, Department of Physiology and Membrane Biology, University of California, Davis, CA |
My research interests and expertise encompass computational structural biology and ion channel biophysics. My research group focuses on the computational design of novel subtype-specific ion channel modulators based on biologics (peptides, proteins, and antibodies) for the treatment of pain, epilepsy, and cardiovascular diseases and on studying molecular determinants of ion channel modulation and gating.
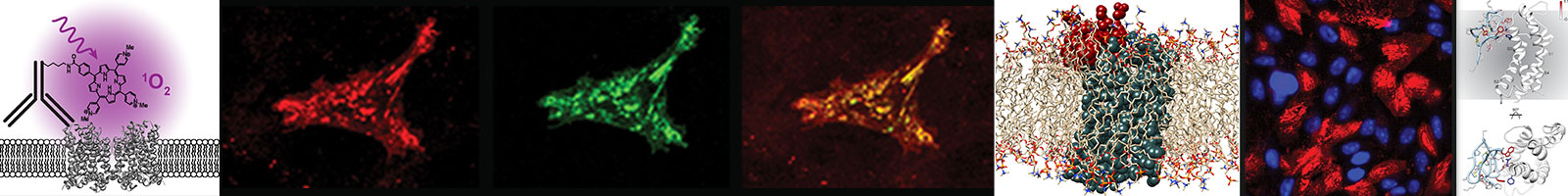
Rational design of novel subtype-specific ion channel modulators. The goal of this project is to optimize potent and selective antibodies and proteins as inhibitors of human voltage-gated sodium (Nav) channels for treatment of chronic pain. Two recent breakthroughs: (1) high-resolution structures revealing human Nav channel interactions with antibodies and peptides, and (2) computational deep learning-based approaches for protein structure prediction and design, have together set the stage for radical optimization of antibodies and peptides as superior therapeutic candidates for the treatment of chronic pain. Pain signals originate predominantly in a subset of peripheral neurons that harbor a distinct subset of Nav channels. Clinical use of Nav channel blockers, such as local anesthetics, is widespread, as they are non-addictive and can provide complete pain relief by inhibiting Nav channels in peripheral sensory neurons. However, local anesthetics are non-selective and also block Nav channels vital for function of the heart, muscle, and central nervous system. One of the key challenges for the development of novel analgesics is selectively targeting Nav channel subtypes involved in pain signaling. Genetic studies have identified human Nav1.7, Nav1.8, and Nav1.9 channel subtypes as key players in pain signaling. These Nav channels are major contributors to action potential generation in peripheral neurons and are established molecular targets for pain therapy. Novel, high-affinity, selective inhibitors of these Nav channels have transformative potential to define a new class of biologics to treat pain.
Ion channel gating and modulation. Function and modulation of Nav channels are critical for excitability in excitable cells. Mutations in human Nav channel genes are responsible for abnormal pain sensation, epilepsy, and cardiovascular diseases. Furthermore, human Nav channels are primary targets of therapeutic drugs used as local anesthetics and for the treatment of neurological and cardiac disorders. This project is focused on studying ion channel modulation by peptides, proteins, and antibodies. The treatment of chronic pain, epilepsy, and cardiovascular diseases remains a major unmet medical need because the use of currently available drugs is limited due to incomplete efficacy and lack of specificity. Considerable efforts by pharmaceutical industry toward identifying selective inhibitors of Nav channel subtypes have not generate genuinely subtype selective blockers. My laboratory uses an innovative approach that combines computational protein structure prediction and whole-cell voltage clamp electrophysiology to study ion channel modulation by peptides, proteins, and antibodies with ultimate goal to design new ion channel modulators. This project will provide key atomic level details on the molecular basis of ion channel gating and modulation that will be essential to design novel therapeutics for treatment of pain, epilepsy, and cardiovascular diseases.
Contemporary Art: |
|
Classical Music: |
|
Contemporary Ballet: |
|
Contemporary Jazz: |
https://www.sfjazz.org/ |
Skiing: |
https://www.skilaketahoe.com/ |
Trans Music: |
https://www.aboveandbeyond.nu/ |
Sports: |
https://www.fcbarcelona.com/en/ |
Wine: |
https://www.napavalley.com/ |
All Rights Reserved | Copyright © Sack and Yarov-Yarovoy Labs