Lab Publications
- 2020
- 2019
- 2018
- 2017
- 2016
- 2015
- 2014
- 2011
- 2010
- 2009
- 2008
2020. Garland MA, Sun B, Zhang S, Reynolds K, Ji Y, Zhou CJ. Role of epigenetics and miRNAs in orofacial clefts. Birth Defects Research (in press)
Abstract. Orofacial clefts (OFCs) have multiple etiologies and likely result from an interplay between genetic and environmental factors. Within the last decade, studies have implicated specific epigenetic modifications and non-coding RNAs as additional facets of OFC etiology. Altered gene expression through DNA methylation and histone modification offer novel insights into how specific genes contribute to distinct OFC subtypes. Epigenetics research has also provided further evidence that cleft lip only (CLO) is a cleft subtype with distinct etiology. Polymorphisms or misexpression of genes encoding microRNAs, as well as their targets, contribute to OFC risk. The ability to experimentally manipulate epigenetic changes and noncoding RNAs in animal models, such as zebrafish, Xenopus, mice, and rats, has offered novel insights into the mechanisms of various OFC subtypes. Although much remains to be understood, recent advancements in our understanding of OFC etiology may advise future strategies of research and preventive care.
2020. Ji Y, Garland MA, Sun B, Zhang S, Reynolds K, McMahon M, Rajakumar R, Islam MS, Liu Y, Chen Y, Zhou CJ. Cellular and developmental basis of orofacial clefts. Birth Defects Research pp1-30.
Abstract. During craniofacial development, defective growth and fusion of the upper lip and/or palate can cause orofacial clefts (OFCs), which are among the most common structural birth defects in humans. The developmental basis of OFCs includes morphogenesis of the upper lip, primary palate, secondary palate, and other orofacial structures, each consisting of diverse cell types originating from all three germ layers: the ectoderm, mesoderm, and endoderm. Cranial neural crest cells and orofacial epithelial cells are two major cell types that interact with various cell lineages and play key roles in orofacial development. The cellular basis of OFCs involves defective execution in any one or several of the following processes: neural crest induction, epithelial-mesenchymal transition, migration, proliferation, differentiation, apoptosis, primary cilia formation and its signaling transduction, epithelial seam formation and disappearance, periderm formation and peeling, convergence and extrusion of palatal epithelial seam cells, cell adhesion, cytoskeleton dynamics, and extracellular matrix function. The latest cellular and developmental findings may provide a basis for better understanding of the underlying genetic, epigenetic, environmental, and molecular mechanisms of OFCs.
2020. Reynolds K, Zhang S, Sun B, Garland MA, Ji Y, Zhou CJ. Genetics and signaling mechanisms of orofacial clefts. Birth Defects Research pp1-47.
Abstract. Craniofacial development involves several complex tissue movements including several fusion processes to form the frontonasal and maxillary structures, including the upper lip and palate. Each of these movements are controlled by many different factors that are tightly regulated by several integral morphogenetic signaling pathways. Subject to both genetic and environmental influences, interruption at nearly any stage can disrupt lip, nasal, or palate fusion and result in a cleft. Here, we discuss many of the genetic risk factors that may contribute to the presentation of orofacial clefts in patients, and several of the key signaling pathways and underlying cellular mechanisms that control lip and palate formation, as identified primarily through investigating equivalent processes in animal models, are examined.
2020. Zhou CJ, Ji Y, Reynolds K, McMahon M, Garland MA, Zhang S, Sun B, Gu R, Islam M, Liu Y, Zhao T, Hsu G, Iwasa J. Non-neural surface ectodermal rosette formation and F-actin dynamics drive mammalian neural tube closure. Biochemical and Biophysical Research Communications 3:647-653.
Highlights
- Non-neural ectodermal cells form multicellular rosettes during mouse posterior neuropore (PNP) closure.
- Rosette-forming cells generate convergent F-actin protrusions and cable network.
- Rosettes and protrusions are disrupted in the surface ectodermal Grhl3-KO mutants.
- Computational modeling visualizes the stepwise closure of mouse PNP.
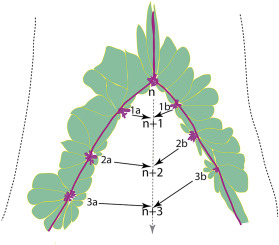 Abstract. The mechanisms underlying mammalian neural tube closure remain poorly understood. We report a unique cellular process involving multicellular rosette formation, convergent cellular protrusions, and F-actin cable network of the non-neural surface ectodermal cells encircling the closure site of the posterior neuropore, which are demonstrated by scanning electron microscopy and genetic fate mapping analyses during mouse spinal neurulation. These unique cellular structures are severely disrupted in the surface ectodermal transcription factor Grhl3 mutants that exhibit fully penetrant spina bifida. We propose a novel model of mammalian neural tube closure driven by surface ectodermal dynamics, which is computationally visualized.
Abstract. The mechanisms underlying mammalian neural tube closure remain poorly understood. We report a unique cellular process involving multicellular rosette formation, convergent cellular protrusions, and F-actin cable network of the non-neural surface ectodermal cells encircling the closure site of the posterior neuropore, which are demonstrated by scanning electron microscopy and genetic fate mapping analyses during mouse spinal neurulation. These unique cellular structures are severely disrupted in the surface ectodermal transcription factor Grhl3 mutants that exhibit fully penetrant spina bifida. We propose a novel model of mammalian neural tube closure driven by surface ectodermal dynamics, which is computationally visualized.
2019. Wang YZ, Fan H, Ji Y, Reynolds K, Gu R, Gan Q, Yamagami T, Zhao T, Hamad S, Bizen N, Takebayashi H, Chen Y, Wu S, Pleasure D, Lam K, Zhou CJ. Olig2 regulates terminal differentiation and maturation of peripheral olfactory sensory neurons. Cellular and Molecular Life Sciences (p1-13)
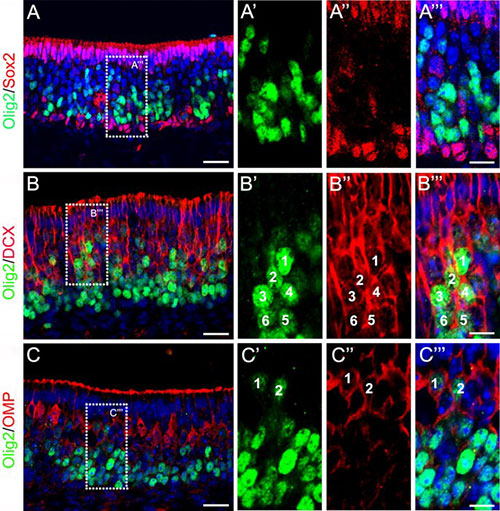 Abstract. The bHLH transcription factor Olig2 is required for sequential cell fate determination of both motor neurons and oligodendrocytes and for progenitor proliferation in the central nervous system. However, the role of Olig2 in peripheral sensory neurogenesis remains unknown. We report that Olig2 is transiently expressed in the newly differentiated olfactory sensory neurons (OSNs) and is down-regulated in the mature OSNs in mice from early gestation to adulthood. Genetic fate mapping demonstrates that Olig2-expressing cells solely give rise to OSNs in the peripheral olfactory system. Olig2 depletion does not affect the proliferation of peripheral olfactory progenitors and the fate determination of OSNs, sustentacular cells, and the olfactory ensheathing cells. However, the terminal differentiation and maturation of OSNs are compromised in either Olig2 single or Olig1/Olig2 double knockout mice, associated with significantly diminished expression of multiple OSN maturation and odorant signaling genes, including Omp, Gnal, Adcy3, and Olfr15. We further demonstrate that Olig2 binds to the E-box in the Omp promoter region to regulate its expression. Taken together, our results reveal a distinctly novel function of Olig2 in the periphery nervous system to regulate the terminal differentiation and maturation of olfactory sensory neurons.
Abstract. The bHLH transcription factor Olig2 is required for sequential cell fate determination of both motor neurons and oligodendrocytes and for progenitor proliferation in the central nervous system. However, the role of Olig2 in peripheral sensory neurogenesis remains unknown. We report that Olig2 is transiently expressed in the newly differentiated olfactory sensory neurons (OSNs) and is down-regulated in the mature OSNs in mice from early gestation to adulthood. Genetic fate mapping demonstrates that Olig2-expressing cells solely give rise to OSNs in the peripheral olfactory system. Olig2 depletion does not affect the proliferation of peripheral olfactory progenitors and the fate determination of OSNs, sustentacular cells, and the olfactory ensheathing cells. However, the terminal differentiation and maturation of OSNs are compromised in either Olig2 single or Olig1/Olig2 double knockout mice, associated with significantly diminished expression of multiple OSN maturation and odorant signaling genes, including Omp, Gnal, Adcy3, and Olfr15. We further demonstrate that Olig2 binds to the E-box in the Omp promoter region to regulate its expression. Taken together, our results reveal a distinctly novel function of Olig2 in the periphery nervous system to regulate the terminal differentiation and maturation of olfactory sensory neurons.
2019. Ji Y, Hao H, Reynolds K, McMahon M, Zhou CJ. Wnt signaling in neural crest ontogenesis and oncogenesis. Cells 8:1173 (p1-40).
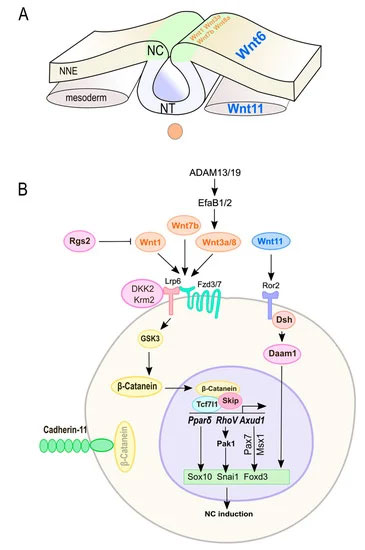
Abstract. Neural crest (NC) cells are a temporary population of multipotent stem cells that generate a diverse array of cell types, including craniofacial bone and cartilage, smooth muscle cells, melanocytes, and peripheral neurons and glia during embryonic development. Defective neural crest development can cause severe and common structural birth defects, such as craniofacial anomalies and congenital heart disease. In the early vertebrate embryos, NC cells emerge from the dorsal edge of the neural tube during neurulation and then migrate extensively throughout the anterior-posterior body axis to generate numerous derivatives. Wnt signaling plays essential roles in embryonic development and cancer. This review summarizes current understanding of Wnt signaling in NC cell induction, delamination, migration, multipotency, and fate determination, as well as in NC-derived cancers.
2019. Kumar S, Reynolds K, Ji Y, Gu R, Rai S, Zhou CJ. Impaired neurodevelopmental pathways in autism spectrum disorder: a review of signaling mechanisms and crosstalk. Journal of Neurodevelopmental Disorders. 11:10 (p1-14)
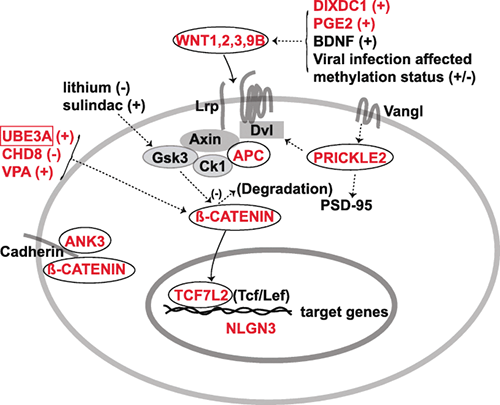 Background: The development of an autistic brain is a highly complex process as evident from the involvement of various genetic and non-genetic factors in the etiology of the autism spectrum disorder (ASD). Despite being a multifactorial neurodevelopmental disorder, autistic patients display a few key characteristics, such as the impaired social interactions and elevated repetitive behaviors, suggesting the perturbation of specific neuronal circuits resulted from abnormal signaling pathways during brain development in ASD. A comprehensive review for autistic signaling mechanisms and interactions may provide a better understanding of ASD etiology and treatment.
Background: The development of an autistic brain is a highly complex process as evident from the involvement of various genetic and non-genetic factors in the etiology of the autism spectrum disorder (ASD). Despite being a multifactorial neurodevelopmental disorder, autistic patients display a few key characteristics, such as the impaired social interactions and elevated repetitive behaviors, suggesting the perturbation of specific neuronal circuits resulted from abnormal signaling pathways during brain development in ASD. A comprehensive review for autistic signaling mechanisms and interactions may provide a better understanding of ASD etiology and treatment.
Main body: Recent studies on genetic models and ASD patients with several different mutated genes revealed the dysregulation of several key signaling pathways, such as WNT, BMP, SHH, and retinoic acid (RA) signaling. Although no direct evidence of dysfunctional FGF or TGF-β signaling in ASD has been reported so far, a few examples of indirect evidence can be found. This review article summarizes how various genetic and non-genetic factors which have been reported contributing to ASD interact with WNT, BMP/TGF-β, SHH, FGF, and RA signaling pathways. The autism-associated gene ubiquitin-protein ligase E3A (UBE3A) has been reported to influence WNT, BMP, and RA signaling pathways, suggesting crosstalk between various signaling pathways during autistic brain development. Finally, the article comments on what further studies could be performed to gain deeper insights into the understanding of perturbed signaling pathways in the etiology of ASD.
Conclusion: The understanding of mechanisms behind various signaling pathways in the etiology of ASD may help to facilitate the identification of potential therapeutic targets and design of new treatment methods.
2019. Reynolds K, Kumari P, Rincon L, Gu R, Ji Y, Kumar S, Zhou CJ. Wnt signaling in orofacial clefts: crosstalk, pathogenesis and models. Disease Models & Mechanisms.12:1-24
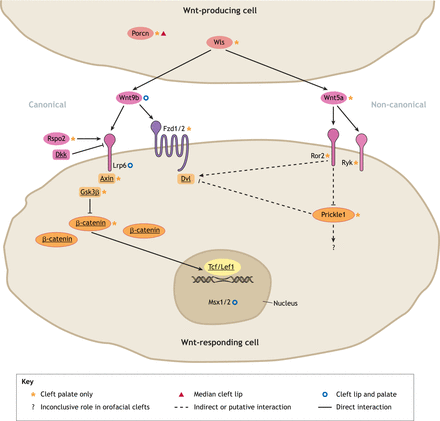 Abstract. Diverse signaling cues and attendant proteins work together during organogenesis, including craniofacial development. Lip and palate formation starts as early as the fourth week of gestation in humans or embryonic day 9.5 in mice. Disruptions in these early events may cause serious consequences, such as orofacial clefts, mainly cleft lip and/or cleft palate. Morphogenetic Wnt signaling, along with other signaling pathways and transcription regulation mechanisms, plays crucial roles during embryonic development, yet the signaling mechanisms and interactions in lip and palate formation and fusion remain poorly understood. Various Wnt signaling and related genes have been associated with orofacial clefts. This Review discusses the role of Wnt signaling and its crosstalk with cell adhesion molecules, transcription factors, epigenetic regulators and other morphogenetic signaling pathways, including the Bmp, Fgf, Tgfβ, Shh and retinoic acid pathways, in orofacial clefts in humans and animal models, which may provide a better understanding of these disorders and could be applied towards prevention and treatments.
Abstract. Diverse signaling cues and attendant proteins work together during organogenesis, including craniofacial development. Lip and palate formation starts as early as the fourth week of gestation in humans or embryonic day 9.5 in mice. Disruptions in these early events may cause serious consequences, such as orofacial clefts, mainly cleft lip and/or cleft palate. Morphogenetic Wnt signaling, along with other signaling pathways and transcription regulation mechanisms, plays crucial roles during embryonic development, yet the signaling mechanisms and interactions in lip and palate formation and fusion remain poorly understood. Various Wnt signaling and related genes have been associated with orofacial clefts. This Review discusses the role of Wnt signaling and its crosstalk with cell adhesion molecules, transcription factors, epigenetic regulators and other morphogenetic signaling pathways, including the Bmp, Fgf, Tgfβ, Shh and retinoic acid pathways, in orofacial clefts in humans and animal models, which may provide a better understanding of these disorders and could be applied towards prevention and treatments.
2018. Fan H, Liu X, Shen Y, Chen S, Huan Y, Shan J, Zhou C, Wu S, Zhang Z, Wang Y. In vivo genetic strategies for the specific lineage tracing of stem cells. Curr Stem Cell Res Ther.
2018. Yamagami T, Pleasure DE, Lam KS, Zhou CJ. Transient activation of Wnt/β-catenin signaling reporter in fibrotic scar formation after compression spinal cord injury in adult mice. Biochemical and Biophysical Research Communications 496:1302-1307.
Highlights
- Wnt signaling reporter TOPgal is transiently activated at the lesion site after compression spinal cord injury (SCI).
- SCI-induced Wnt signaling activities appeared in day 5, peaked in day 7, and diminished in day 14 post injury.
- SCI-induced TOPgal positive cells are fibrotic cells in the core score region.
- SCI-induced TOPgal positive fibroblast-like cells are proliferative.
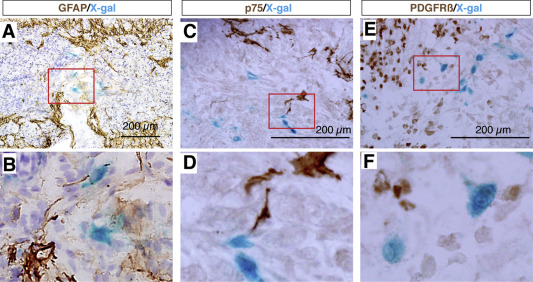
Abstract. After traumatic spinal cord injury (SCI), a scar may form with a fibrotic core (fibrotic scar) and surrounding reactive astrocytes (glial scar) at the lesion site. The scar tissue is considered a major obstacle preventing regeneration both as a physical barrier and as a source for secretion of inhibitors of axonal regeneration. Understanding the mechanism of scar formation and how to control it may lead to effective SCI therapies. Using a compression-SCI model on adult transgenic mice, we demonstrate that the canonical Wnt/β-catenin signaling reporter TOPgal (TCF/Lef1-lacZ) positive cells appeared at the lesion site by 5 days, peaked on 7 days, and diminished by 14 days post injury. Using various representative cell lineage markers, we demonstrate that, these transiently TOPgal positive cells are a group of Fibronectin(+); GFAP(−) fibroblast-like cells in the core scar region. Some of them are proliferative. These results indicate that Wnt/β-catenin signaling may play a key role in fibrotic scar formation after traumatic spinal cord injury.
2018. Wang Y, Zhou CJ, Liu Y. Wnt signaling in kidney development and disease. Progress in Molecular Biology and Translational Science 153:181-207.
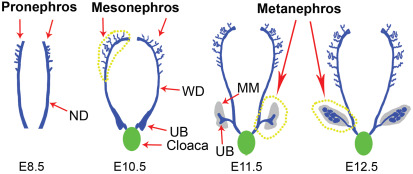 Abstract. Wnt signal cascade is an evolutionarily conserved, developmental pathway that regulates embryogenesis, injury repair, and pathogenesis of human diseases. It is well established that Wnt ligands transmit their signal via canonical, β-catenin-dependent and noncanonical, β-catenin-independent mechanisms. Mounting evidence has revealed that Wnt signaling plays a key role in controlling early nephrogenesis and is implicated in the development of various kidney disorders. Dysregulations of Wnt expression cause a variety of developmental abnormalities and human diseases, such as congenital anomalies of the kidney and urinary tract, cystic kidney, and renal carcinoma. Multiple Wnt ligands, their receptors, and transcriptional targets are upregulated during nephron formation, which is crucial for mediating the reciprocal interaction between primordial tissues of ureteric bud and metanephric mesenchyme. Renal cysts are also associated with disrupted Wnt signaling. In addition, Wnt components are important players in renal tumorigenesis. Activation of Wnt/β-catenin is instrumental for tubular repair and regeneration after acute kidney injury. However, sustained activation of this signal cascade is linked to chronic kidney diseases and renal fibrosis in patients and experimental animal models. Mechanistically, Wnt signaling controls a diverse array of biologic processes, such as cell cycle progression, cell polarity and migration, cilia biology, and activation of renin–angiotensin system. In this chapter, we have reviewed recent findings that implicate Wnt signaling in kidney development and diseases. Targeting this signaling may hold promise for future treatment of kidney disorders in patients.
Abstract. Wnt signal cascade is an evolutionarily conserved, developmental pathway that regulates embryogenesis, injury repair, and pathogenesis of human diseases. It is well established that Wnt ligands transmit their signal via canonical, β-catenin-dependent and noncanonical, β-catenin-independent mechanisms. Mounting evidence has revealed that Wnt signaling plays a key role in controlling early nephrogenesis and is implicated in the development of various kidney disorders. Dysregulations of Wnt expression cause a variety of developmental abnormalities and human diseases, such as congenital anomalies of the kidney and urinary tract, cystic kidney, and renal carcinoma. Multiple Wnt ligands, their receptors, and transcriptional targets are upregulated during nephron formation, which is crucial for mediating the reciprocal interaction between primordial tissues of ureteric bud and metanephric mesenchyme. Renal cysts are also associated with disrupted Wnt signaling. In addition, Wnt components are important players in renal tumorigenesis. Activation of Wnt/β-catenin is instrumental for tubular repair and regeneration after acute kidney injury. However, sustained activation of this signal cascade is linked to chronic kidney diseases and renal fibrosis in patients and experimental animal models. Mechanistically, Wnt signaling controls a diverse array of biologic processes, such as cell cycle progression, cell polarity and migration, cilia biology, and activation of renin–angiotensin system. In this chapter, we have reviewed recent findings that implicate Wnt signaling in kidney development and diseases. Targeting this signaling may hold promise for future treatment of kidney disorders in patients.
2017. Huang C, Chen Y, Liu H, Jing Yang, Song X, Zhao J, Na He N, Zhou CJ, Wang Y, Huang C, Dong Q. Celecoxib targets breast cancer stem cells by inhibiting the synthesis of prostaglandin E2 and down-regulating the Wnt pathway activity. Oncotarget 8:115254-115269.
Abstract. Pharmacological targeting of breast cancer stem cells (CSCs) is highly promising for the treatment of breast cancer, as the small population of CSCs is responsible for tumor initiation, progression, recurrence and chemo-resistance. Celecoxib is one of the most commonly used non-steroidal anti-inflammatory drugs (NSAIDs), which have chemo-preventive activity against cancers, including breast cancer and colorectal cancer. However, the mechanisms by which NSAIDs exert its cancer prevention effects have yet been completely understood. In the present study, we investigated for the first time the effect of celecoxib on breast CSCs inhibition and its potential molecular mechanisms. Our results demonstrated that celecoxib suppresses CSC self-renewal, sensitizes chemo-resistance, inhibits epithelial to mesenchymal transition (EMT), and attenuates metastasis and tumorigenesis. Further exploring the underlying mechanism revealed that celecoxib targets breast CSCs by inhibiting the synthesis of prostaglandin E2 and down-regulating the Wnt pathway activity. Our findings suggest that celecoxib, by targeting CSCs, may be used as an adjuvant chemotherapy drug to improve breast cancer treatment outcomes.
2016. Dong Z, Huang M, Liu Z, Xie P, Dong Y, Wu X, Qu Z, Shen B, Huang X, Zhang T, Li J, Liu J, Yanase T, Zhou C, Xu Y. Focused screening of mitochondrial metabolism reveals a crucial role for a tumor suppressor Hbp1 in ovarian reserve. Cell Death and Differentiation 23:1602-1614.
Abstract: Granulosa cells (GCs) are tightly associated with fertility and the fate of ovarian follicles. Mitochondria are the central executers of apoptosis. However, the genetic basis underlying mitochondrial modulation in GCs during the ovarian development is poorly understood. Here, CRISPR/Cas9-mediated genetic screening was used to identify genes conferring mitochondrial metabolism in human GCs. The results uncovered roles for several tumor suppressors, including HBP1, in the augmentation of mitochondrial function. Focused analysis revealed that high-mobility group (HMG)-box transcription factor 1 (Hbp1) levels regulate mitochondrial biogenesis, which is associated with global changes in transcription including Tfam. The systemic or granulosa-specific but not oocyte-specific ablation of Hbp1promoted follicle growth and oocyte production, and is associated with the reduced apoptotic signals in mouse GCs. Consistent with increased mitochondrial function and attenuated GC apoptosis, the regulation of Hbp1 conferred substantial protection of ovarian reserve. Thus, the results of the present study provide a critical target to understand the control of the reproductive lifespan. Cell Death and Differentiation advance online publication, 20 May 2016; doi:10.1038/cdd.2016.47.
2015. Wang Y, Stokes A, Duan Z, Hui J, Xu Y, Chen YP, Chen HW, Lam K, Zhou CJ. Lrp6 modulates Ret signaling in renal development and cystic dysplasia. Journal of the American Society of Nephrology 27:417-427. DOI: 10.1681/ASN.2014100998
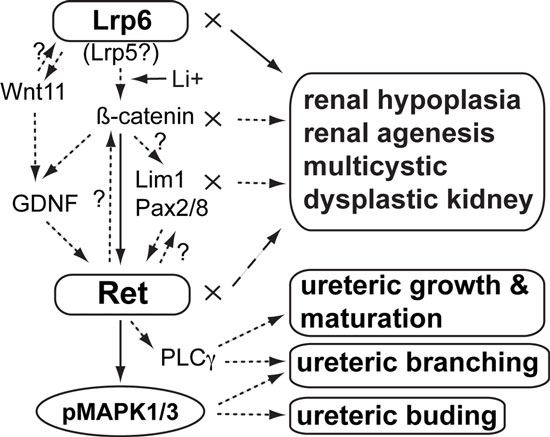 Abstract: Hypoplastic and/or cystic kidneys have been found in both LDL receptor–related protein 6 (Lrp6)- and β-catenin–mutant mouse embryos, and these proteins are key molecules for Wnt signaling. However, the underlying mechanisms of Lrp6/β-catenin signaling in renal development and cystic formation remain poorly understood. In this study, we found evidence that diminished cell proliferation and increased apoptosis occur before cystic dysplasia in the renal primordia of Lrp6-deficient mouse embryos. The expression of Ret proto-oncogene (Ret), a critical receptor for the growth factor glial cell line–derived neurotrophic factor (GDNF), which is required for early nephrogenesis, was dramatically diminished in the mutant renal primordia. The activities of other representative nephrogenic genes, including Lim1, Pax2, Pax8, GDNF, and Wnt11, were subsequently diminished in the mutant renal primordia. Molecular biology experiments demonstrated that Ret is a novel transcriptional target of Wnt/β-catenin signaling. Wnt agonist lithium promoted Ret expression in vitro and in vivo. Furthermore, Lrp6-knockdown or lithium treatment in vitro led to downregulation or upregulation, respectively, of the phosphorylated mitogen-activated protein kinases 1 and 3, which act downstream of GDNF/Ret signaling. Mice with single and double mutations of Lrp6 and Ret were perinatal lethal and demonstrated gene dosage–dependent effects on the severity of renal hypoplasia during embryogenesis. Taken together, these results suggest that Lrp6-mediated Wnt/β-catenin signaling modulates or interacts with a signaling network consisting of Ret cascades and related nephrogenic factors for renal development, and the disruption of these genes or signaling activities may cause a spectrum of hypoplastic and cystic kidney disorders.
Abstract: Hypoplastic and/or cystic kidneys have been found in both LDL receptor–related protein 6 (Lrp6)- and β-catenin–mutant mouse embryos, and these proteins are key molecules for Wnt signaling. However, the underlying mechanisms of Lrp6/β-catenin signaling in renal development and cystic formation remain poorly understood. In this study, we found evidence that diminished cell proliferation and increased apoptosis occur before cystic dysplasia in the renal primordia of Lrp6-deficient mouse embryos. The expression of Ret proto-oncogene (Ret), a critical receptor for the growth factor glial cell line–derived neurotrophic factor (GDNF), which is required for early nephrogenesis, was dramatically diminished in the mutant renal primordia. The activities of other representative nephrogenic genes, including Lim1, Pax2, Pax8, GDNF, and Wnt11, were subsequently diminished in the mutant renal primordia. Molecular biology experiments demonstrated that Ret is a novel transcriptional target of Wnt/β-catenin signaling. Wnt agonist lithium promoted Ret expression in vitro and in vivo. Furthermore, Lrp6-knockdown or lithium treatment in vitro led to downregulation or upregulation, respectively, of the phosphorylated mitogen-activated protein kinases 1 and 3, which act downstream of GDNF/Ret signaling. Mice with single and double mutations of Lrp6 and Ret were perinatal lethal and demonstrated gene dosage–dependent effects on the severity of renal hypoplasia during embryogenesis. Taken together, these results suggest that Lrp6-mediated Wnt/β-catenin signaling modulates or interacts with a signaling network consisting of Ret cascades and related nephrogenic factors for renal development, and the disruption of these genes or signaling activities may cause a spectrum of hypoplastic and cystic kidney disorders.
2014. Zhao T*, Gan Q*, Stokes A, Lassiter RN, Wang YP, Chan J, Han J, Pleasure DE, Epstein JA, Zhou CJ. Beta-catenin regulates Pax3 and Cdx2 for caudal neural tube closure and elongation. Development 141:148-157. DOI: 10.1242/dev.101550
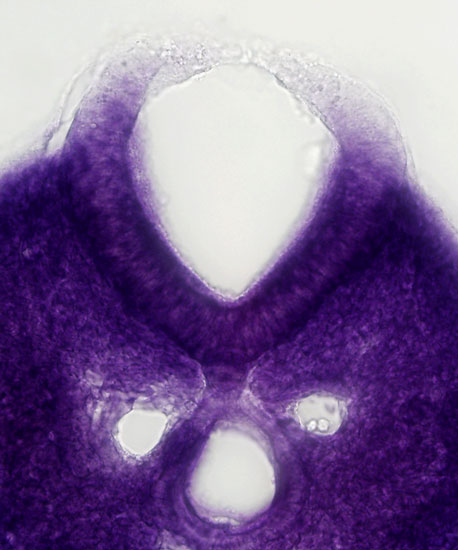 Abstract: Non-canonical Wnt/planar cell polarity (PCP) signaling plays a primary role in the convergent extension that drives neural tube closure and body axis elongation. PCP signaling gene mutations cause severe neural tube defects (NTDs). However, the role of canonical Wnt/β-catenin signaling in neural tube closure and NTDs remains poorly understood. This study shows that conditional gene targeting of β-catenin in the dorsal neural folds of mouse embryos represses the expression of the homeobox-containing genes Pax3 and Cdx2 at the dorsal posterior neuropore (PNP), and subsequently diminishes the expression of the Wnt/β-catenin signaling target genes T, Tbx6 and Fgf8 at the tail bud, leading to spina bifida aperta, caudal axis bending and tail truncation. We demonstrate that Pax3 and Cdx2 are novel downstream targets of Wnt/β-catenin signaling. Transgenic activation of Pax3 cDNA can rescue the closure defect in the β-catenin mutants, suggesting that Pax3 is a key downstream effector of β-catenin signaling in the PNP closure process. Cdx2 is known to be crucial in posterior axis elongation and in neural tube closure. We found that Cdx2 expression is also repressed in the dorsal PNPs of Pax3-null embryos. However, the ectopically activated Pax3 in the β-catenin mutants cannot restore Cdx2 mRNA in the dorsal PNP, suggesting that the presence of both β-catenin and Pax3 is required for regional Cdx2 expression. Thus, β-catenin signaling is required for caudal neural tube closure and elongation, acting through the transcriptional regulation of key target genes in the PNP.
Abstract: Non-canonical Wnt/planar cell polarity (PCP) signaling plays a primary role in the convergent extension that drives neural tube closure and body axis elongation. PCP signaling gene mutations cause severe neural tube defects (NTDs). However, the role of canonical Wnt/β-catenin signaling in neural tube closure and NTDs remains poorly understood. This study shows that conditional gene targeting of β-catenin in the dorsal neural folds of mouse embryos represses the expression of the homeobox-containing genes Pax3 and Cdx2 at the dorsal posterior neuropore (PNP), and subsequently diminishes the expression of the Wnt/β-catenin signaling target genes T, Tbx6 and Fgf8 at the tail bud, leading to spina bifida aperta, caudal axis bending and tail truncation. We demonstrate that Pax3 and Cdx2 are novel downstream targets of Wnt/β-catenin signaling. Transgenic activation of Pax3 cDNA can rescue the closure defect in the β-catenin mutants, suggesting that Pax3 is a key downstream effector of β-catenin signaling in the PNP closure process. Cdx2 is known to be crucial in posterior axis elongation and in neural tube closure. We found that Cdx2 expression is also repressed in the dorsal PNPs of Pax3-null embryos. However, the ectopically activated Pax3 in the β-catenin mutants cannot restore Cdx2 mRNA in the dorsal PNP, suggesting that the presence of both β-catenin and Pax3 is required for regional Cdx2 expression. Thus, β-catenin signaling is required for caudal neural tube closure and elongation, acting through the transcriptional regulation of key target genes in the PNP.
2014. Gan Q*, Lee A*, Suzuki R*, Yamagami T, Stokes A, Nguyen B, Pleasure D, Wang J, Chen HW, Zhou CJ. Pax6 mediates ß-catenin signaling for self-renewal and neurogenesis by neocortical radial glial stem cells. Stem Cells 32:45-58. DOI: 10.1002/stem.1561
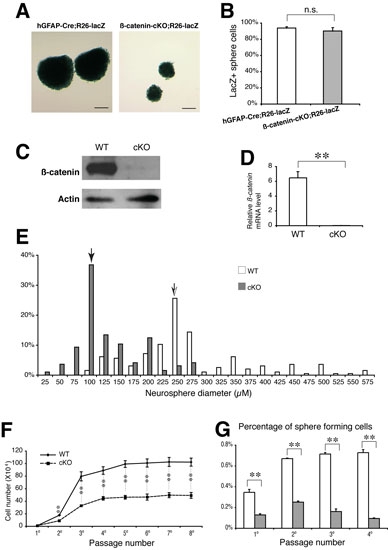 Abstract: The Wnt/ß-catenin pathway is a critical stem cell regulator and plays important roles in neuroepithelial cells during early gestation. However, the role of Wnt/ß-catenin signaling in radial glia, a major neural stem cell population expanded by mid-gestation, remains poorly understood. This study shows that genetic ablation of ß-catenin with hGFAP-Cre mice inhibits neocortical formation by disrupting radial glial development. Reduced radial glia and intermediate progenitors are found in the ß-catenin-deficient neocortex during late gestation. Increased apoptosis and divergent localization of radial glia in the subventricular zone are also observed in the mutant neocortex. In vivo and in vitro proliferation and neurogenesis as well as oligodendrogenesis by cortical radial glia or by dissociated neural stem cells are significantly defective in the mutants. Neocortical layer patterning is not apparently altered, while astrogliogenesis is ectopically increased in the mutants. At the molecular level, the expression of the transcription factor Pax6 is dramatically diminished in the cortical radial glia and the sphere-forming neural stem cells of ß-catenin-deficient mutants. Chromatin immunoprecipitation and luciferase assays demonstrate that ß-catenin/Tcf complex binds to Pax6 promoter and induces its transcriptional activities. The forced expression of Pax6 through lentiviral transduction partially rescues the defective proliferation and neurogenesis by ß-catenin-deficient neural stem cells. Thus, Pax6 is a novel downstream target of the Wnt/ß-catenin pathway, and ß-catenin/Pax6 signaling plays critical roles in self-renewal and neurogenesis of radial glia/neural stem cells during neocortical development.
Abstract: The Wnt/ß-catenin pathway is a critical stem cell regulator and plays important roles in neuroepithelial cells during early gestation. However, the role of Wnt/ß-catenin signaling in radial glia, a major neural stem cell population expanded by mid-gestation, remains poorly understood. This study shows that genetic ablation of ß-catenin with hGFAP-Cre mice inhibits neocortical formation by disrupting radial glial development. Reduced radial glia and intermediate progenitors are found in the ß-catenin-deficient neocortex during late gestation. Increased apoptosis and divergent localization of radial glia in the subventricular zone are also observed in the mutant neocortex. In vivo and in vitro proliferation and neurogenesis as well as oligodendrogenesis by cortical radial glia or by dissociated neural stem cells are significantly defective in the mutants. Neocortical layer patterning is not apparently altered, while astrogliogenesis is ectopically increased in the mutants. At the molecular level, the expression of the transcription factor Pax6 is dramatically diminished in the cortical radial glia and the sphere-forming neural stem cells of ß-catenin-deficient mutants. Chromatin immunoprecipitation and luciferase assays demonstrate that ß-catenin/Tcf complex binds to Pax6 promoter and induces its transcriptional activities. The forced expression of Pax6 through lentiviral transduction partially rescues the defective proliferation and neurogenesis by ß-catenin-deficient neural stem cells. Thus, Pax6 is a novel downstream target of the Wnt/ß-catenin pathway, and ß-catenin/Pax6 signaling plays critical roles in self-renewal and neurogenesis of radial glia/neural stem cells during neocortical development.
2014. Lassiter RN, Stark MR, Zhao T, Zhou CJ. Signaling mechanisms controlling cranial placode neurogenesis and delamination. Developmental Biology 389:39-49. DOI: 10.1016/j.ydbio.2013.11.025
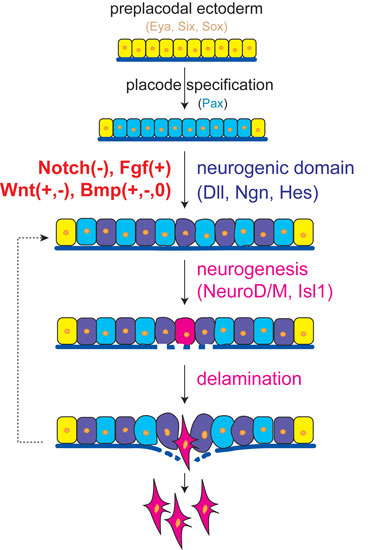 Highlights
Highlights
• A core signaling program exists throughout the neurogenic placodes.
• Attenuated Notch signaling initiates placode neurogenesis and delamination.
• Active FGF signaling also triggers placode neurogenesis and delamination.
• Wnt and BMP pathways exert context-dependent roles in neurogenic placodes.
Abstract: The neurogenic cranial placodes are a unique transient epithelial niche of neural progenitor cells that give rise to multiple derivatives of the peripheral nervous system, particularly, the sensory neurons. Placode neurogenesis occurs throughout an extended period of time with epithelial cells continually recruited as neural progenitor cells. Sensory neuron development in the trigeminal, epibranchial, otic, and olfactory placodes coincides with detachment of these neuroblasts from the encompassing epithelial sheet, leading to delamination and ingression into the mesenchyme where they continue to differentiate as neurons. Multiple signaling pathways are known to direct placodal development. This review defines the signaling pathways working at the finite spatiotemporal period when neuronal selection within the placodes occurs, and neuroblasts concomitantly delaminate from the epithelium. Examining neurogenesis and delamination after initial placodal patterning and specification has revealed a common trend throughout the neurogenic placodes, which suggests that both activated FGF and attenuated Notch signaling activities are required for neurogenesis and changes in epithelial cell adhesion leading to delamination. We also address the varying roles of other pathways such as the Wnt and BMP signaling families during sensory neurogenesis and neuroblast delamination in the differing placodes.
2011. Wang Y, Song L, Zhou CJ. The canonical Wnt/beta-catenin signaling pathway regulates Fgf signaling for early facial development. Developmental Biology 349:250-260.
 Abstract: The canonical Wnt/β-catenin signaling pathway has implications in early facial development; yet, its function and signaling mechanism remain poorly understood. We report here that the frontonasal and upper jaw primordia cannot be formed after conditional ablation of β-catenin with Foxg1-Cre mice in the facial ectoderm and the adjacent telencephalic neuroepithelium. Gene expression of several cell-survival and patterning factors, including Fgf8, Fgf3, and Fgf17, is dramatically diminished in the anterior neural ridge (ANR, a rostral signaling center) and/or the adjacent frontonasal ectoderm of the β-catenin conditional mutant mice. In addition, Shh expression is diminished in the ventral telencephalon of the mutants, while Tcfap2a expression is less affected in the facial primordia. Apoptosis occurs robustly in the rostral head tissues following inactivation of Fgf signaling in the conditional mutants. Consequently, the upper jaw, nasal, ocular and telencephalic structures are absent, but the tongue and mandible are relatively developed in the conditional mutants at birth. Using molecular biological approaches, we demonstrate that the Fgf8 gene is transcriptionally targeted by Wnt/β-catenin signaling during early facial and forebrain development. Furthermore, we show that conditional gain-of-function of β-catenin signaling causes drastic upregulation of Fgf8 mRNA in the ANR and the entire facial ectoderm, which also arrests facial and forebrain development. Taken together, our results suggest that canonical Wnt/β-catenin signaling is required for early development of the mammalian face and related head structures, which mainly or partly acts through the initiation and modulation of balanced Fgf signaling activity.
Abstract: The canonical Wnt/β-catenin signaling pathway has implications in early facial development; yet, its function and signaling mechanism remain poorly understood. We report here that the frontonasal and upper jaw primordia cannot be formed after conditional ablation of β-catenin with Foxg1-Cre mice in the facial ectoderm and the adjacent telencephalic neuroepithelium. Gene expression of several cell-survival and patterning factors, including Fgf8, Fgf3, and Fgf17, is dramatically diminished in the anterior neural ridge (ANR, a rostral signaling center) and/or the adjacent frontonasal ectoderm of the β-catenin conditional mutant mice. In addition, Shh expression is diminished in the ventral telencephalon of the mutants, while Tcfap2a expression is less affected in the facial primordia. Apoptosis occurs robustly in the rostral head tissues following inactivation of Fgf signaling in the conditional mutants. Consequently, the upper jaw, nasal, ocular and telencephalic structures are absent, but the tongue and mandible are relatively developed in the conditional mutants at birth. Using molecular biological approaches, we demonstrate that the Fgf8 gene is transcriptionally targeted by Wnt/β-catenin signaling during early facial and forebrain development. Furthermore, we show that conditional gain-of-function of β-catenin signaling causes drastic upregulation of Fgf8 mRNA in the ANR and the entire facial ectoderm, which also arrests facial and forebrain development. Taken together, our results suggest that canonical Wnt/β-catenin signaling is required for early development of the mammalian face and related head structures, which mainly or partly acts through the initiation and modulation of balanced Fgf signaling activity.
2011. He F, Xiong W, Wang Y, Li L, Liu C, Yamagami T, Taketo MM, Zhou CJ, Chen Y. Epithelial Wnt/ß-catenin signaling regulates palatal shelf fusion through regulation of Tgfß3 expression. Developmental Biology 350:511-519. (Collaborative contribution to the Chen Lab's revision experiment)
Abstract: The canonical Wnt/β-catenin signaling plays essential role in development and diseases. Previous studies have implicated the canonical Wnt/β-catenin signaling in the regulation of normal palate development, but functional Wnt/β-catenin signaling and its tissue-specific activities remain to be accurately elucidated. In this study, we show that functional Wnt/β-catenin signaling operates primarily in the palate epithelium, particularly in the medial edge epithelium (MEE) of the developing mouse palatal shelves, consistent with the expression patterns of β-catenin and several Wnt ligands and receptors. Epithelial specific inactivation of β-catenin by the K14-Cre transgenic allele abolishes the canonical Wnt signaling activity in the palatal epithelium and leads to an abnormal persistence of the medial edge seam (MES), ultimately causing a cleft palate formation, a phenotype resembling that in Tgfβ3 mutant mice. Consistent with this phenotype is the down-regulation of Tgfβ3 and suppression of apoptosis in the MEE of the β-catenin mutant palatal shelves. Application of exogenous Tgfβ3 to the mutant palatal shelves in organ culture rescues the midline seam phenotype. On the other hand, expression of stabilized β-catenin in the palatal epithelium also disrupts normal palatogenesis by activating ectopic Tgfβ3 expression in the palatal epithelium and causing an aberrant fusion between the palate shelf and mandible in addition to severely deformed palatal shelves. Collectively, our results demonstrate an essential role for Wnt/β-catenin signaling in the epithelial component at the step of palate fusion during palate development by controlling the expression of Tgfβ3 in the MEE.
2011. Wang YZ, Yamagami T, Gan Q, Wang Y, Zhao T, Hamad S, Lott P, Schnittke N, Schwob JE, Zhou CJ. Canonical Wnt signaling promotes proliferation and neurogenesis of peripheral olfactory stem cells during postnatal development and adult regeneration. Journal of Cell Science 124:1553-1563. (journal cover)
 Abstract: The mammalian olfactory epithelium (OE) has a unique stem cell or progenitor niche, which is responsible for the constant peripheral neurogenesis throughout the lifespan of the animal. However, neither the signals that regulate the behavior of these cells nor the lineage properties of the OE stem cells are well understood. Multiple Wnt signaling components exhibit dynamic expression patterns in the developing OE. We generated Wnt signaling reporter TOPeGFP transgenic mice and found TOPeGFP activation predominantly in proliferating Sox2(+) OE basal cells during early postnatal development. FACS-isolated TOPeGFP(+) OE basal cells are required, but are not sufficient, for formation of spheres. Wnt3a significantly promotes the proliferation of the Sox2(+) OE sphere cells. Wnt-stimulated OE sphere cells maintain their multipotency and can differentiate into most types of neuronal and non-neuronal epithelial cells. Also, Wnt activators shift the production of differentiated cells toward olfactory sensory neurons. Moreover, TOPeGFP(+) cells are robustly increased in the adult OE after injury. In vivo administration of Wnt modulators significantly alters the regeneration potential. This study demonstrates the role of the canonical Wnt signaling pathway in the regulation of OE stem cells or progenitors during development and regeneration.
Abstract: The mammalian olfactory epithelium (OE) has a unique stem cell or progenitor niche, which is responsible for the constant peripheral neurogenesis throughout the lifespan of the animal. However, neither the signals that regulate the behavior of these cells nor the lineage properties of the OE stem cells are well understood. Multiple Wnt signaling components exhibit dynamic expression patterns in the developing OE. We generated Wnt signaling reporter TOPeGFP transgenic mice and found TOPeGFP activation predominantly in proliferating Sox2(+) OE basal cells during early postnatal development. FACS-isolated TOPeGFP(+) OE basal cells are required, but are not sufficient, for formation of spheres. Wnt3a significantly promotes the proliferation of the Sox2(+) OE sphere cells. Wnt-stimulated OE sphere cells maintain their multipotency and can differentiate into most types of neuronal and non-neuronal epithelial cells. Also, Wnt activators shift the production of differentiated cells toward olfactory sensory neurons. Moreover, TOPeGFP(+) cells are robustly increased in the adult OE after injury. In vivo administration of Wnt modulators significantly alters the regeneration potential. This study demonstrates the role of the canonical Wnt signaling pathway in the regulation of OE stem cells or progenitors during development and regeneration.
Cover caption: The newly constructed Wnt signalling reporter TOPeGFP (green) is predominantly activated in the olfactory epithelial basal stem cells in early postnatal transgenic mice. Olfactory marker protein immunolabelling (red) shows the bipolar olfactory receptor neurons with extended dendrites towards the lumenal surface and axon bundles distributed in the lamina propria. See article by Wang et al. (pp. 1553-1563).
2011. Wang YZ, Plane JM, Jiang P, Zhou CJ, Deng W. Concise Review: Quiescent and active states of endogenous adult neural stem cells: identification and characterization. Stem Cells 29:907-912.
Abstract: The adult mammalian central nervous system (CNS) lacks the capacity for regeneration, making it a highly sought-after topic for researchers. The identification of neural stem cells (NSCs) in the adult CNS wiped out a long-held dogma that the adult brain contains a set number of neurons and is incapable of replacing them. The discovery of adult NSCs (aNSCs) stoked the fire for researchers who dream of brain self-repair. Unfortunately, the quiescent nature and limited plasticity of aNSCs diminish their regenerative potential. Recent studies evaluating aNSC plasticity under pathological conditions indicate that a switch from quiescent to active aNSCs in neurogenic regions plays an important role in both repairing the damaged tissue and preserving progenitor pools. Here, we summarize the most recent findings and present questions about characterizing the active and quiescent aNSCs in major neurogenic regions, and factors for maintaining their active and quiescent states, hoping to outline an emerging view for promoting the endogenous aNSC-based regeneration.
2011. Guo F, Maeda Y, Ma J, Delgado M, Sohn J, Miers L, Ko EM, Bannerman, Xu J, Wang Y, Zhou CJ, Takebayashi H, Pleasure D. Macroglial plasticity and the origins of reactive astroglia in experimental autoimmune encephalomyelitis. Journal of Neuroscience 31:11914-11928.
Abstract: Accumulations of hypertrophic, intensely glial fibrillary acidic protein-positive (GFAP(+)) astroglia, which also express immunoreactive nestin and vimentin, are prominent features of multiple sclerosis lesions. The issues of the cellular origin of hypertrophic GFAP(+)/vimentin(+)/nestin(+) "reactive" astroglia and also the plasticities and lineage relationships among three macroglial progenitor populations-oligodendrocyte progenitor cells (OPCs), astrocytes and ependymal cells-during multiple sclerosis and other CNS diseases remain controversial. We used genetic fate-mappings with a battery of inducible Cre drivers (Olig2-Cre-ER(T2), GFAP-Cre-ER(T2), FoxJ1-Cre-ER(T2) and Nestin-Cre-ER(T2)) to explore these issues in adult mice with myelin oligodendrocyte glycoprotein peptide-induced experimental autoimmune encephalomyelitis (EAE). The proliferative rate of spinal cord OPCs rose fivefold above control levels during EAE, and numbers of oligodendroglia increased as well, but astrogenesis from OPCs was rare. Spinal cord ependymal cells, previously reported to be multipotent, did not augment their low proliferative rate, nor give rise to astroglia or OPCs. Instead, the hypertrophic, vimentin(+)/nestin(+), reactive astroglia that accumulated in spinal cord in this multiple sclerosis model were derived by proliferation and phenotypic transformation of fibrous astroglia in white matter, and solely by phenotypic transformation of protoplasmic astroglia in gray matter. This comprehensive analysis of macroglial plasticity in EAE helps to clarify the origins of astrogliosis in CNS inflammatory demyelinative disorders.
2010. Zhou CJ, Wang YZ, Yamagami T, Zhao T, Song L, Wang K. Generation of Lrp6 conditional gene-targeting mouse line for modeling and dissecting multiple birth defects/congenital anomalies. Developmental Dynamics 239:318-326.
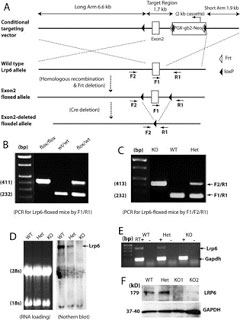 Abstract: Lrp6 is a key coreceptor in the canonical Wnt pathway that is widely involved in tissue/organ morphogenesis. We generated a loxP-floxed Lrp6 mouse line. Crossing with a general Cre deleter, we obtained the Lrp6-floxdel mice, in which the loxP-floxed exon 2 of Lrp6 gene has been deleted ubiquitously. The homozygotes of Lrp6-floxdel mice reproduced typical defects as seen in the conventional Lrp6-deficient mice, such as defects in eye, limb, and neural tube, and die around birth. We also found new phenotypes including cleft palate and agenesis of external genitalia in the Lrp6-floxdel mice. In addition, the Lrp6-deficient embryos are known to be defective in other systems and internal organs including the heart and brain. Thus, by selectively crossing with a lineage-specific or inducible Cre mouse line, the Lrp6 conditional gene-targeting mice will allow us to model specific types of birth defects for mechanism and prevention studies.
Abstract: Lrp6 is a key coreceptor in the canonical Wnt pathway that is widely involved in tissue/organ morphogenesis. We generated a loxP-floxed Lrp6 mouse line. Crossing with a general Cre deleter, we obtained the Lrp6-floxdel mice, in which the loxP-floxed exon 2 of Lrp6 gene has been deleted ubiquitously. The homozygotes of Lrp6-floxdel mice reproduced typical defects as seen in the conventional Lrp6-deficient mice, such as defects in eye, limb, and neural tube, and die around birth. We also found new phenotypes including cleft palate and agenesis of external genitalia in the Lrp6-floxdel mice. In addition, the Lrp6-deficient embryos are known to be defective in other systems and internal organs including the heart and brain. Thus, by selectively crossing with a lineage-specific or inducible Cre mouse line, the Lrp6 conditional gene-targeting mice will allow us to model specific types of birth defects for mechanism and prevention studies.
2010. Song L, Li Y, Wang K, Zhou CJ. Cardiac neural crest and outflow tract defects in Lrp6 mutant mice. Developmental Dynamics 239:200-210.
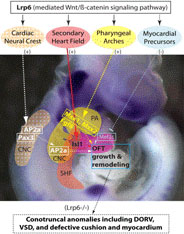 Abstract: The role of a key Wnt coreceptor Lrp6 during heart development remains unclear. Here we show that ablation of Lrp6 in mice causes conotruncal anomalies including double-outlet right ventricle (DORV), outflow tract (OFT) cushion hypoplasia, and ventricular septal defect (VSD). Cardiac neural crest cells are specifically lost in the dorsal neural tube and caudal pharyngeal arches of the mutant embryos. We also demonstrate that Lrp6 is required for proliferation and survival of cardiac progenitors and for the expression of Isl1 in the secondary heart field. Other known cardiogenic regulators such as Msx1, Msx2, and Fgf8 are also significantly diminished in the mutant pharyngeal arches and/or OFT. Unexpectedly, the myocardium differentiation factors Mef2c and Myocardin are upregulated in the mutant OFT. Our results indicate that Lrp6 is essential for cardiac neural crest and OFT development upstream of multiple important cardiogenic genes in different cardiac lineage cells during early cardiogenesis.
Abstract: The role of a key Wnt coreceptor Lrp6 during heart development remains unclear. Here we show that ablation of Lrp6 in mice causes conotruncal anomalies including double-outlet right ventricle (DORV), outflow tract (OFT) cushion hypoplasia, and ventricular septal defect (VSD). Cardiac neural crest cells are specifically lost in the dorsal neural tube and caudal pharyngeal arches of the mutant embryos. We also demonstrate that Lrp6 is required for proliferation and survival of cardiac progenitors and for the expression of Isl1 in the secondary heart field. Other known cardiogenic regulators such as Msx1, Msx2, and Fgf8 are also significantly diminished in the mutant pharyngeal arches and/or OFT. Unexpectedly, the myocardium differentiation factors Mef2c and Myocardin are upregulated in the mutant OFT. Our results indicate that Lrp6 is essential for cardiac neural crest and OFT development upstream of multiple important cardiogenic genes in different cardiac lineage cells during early cardiogenesis.
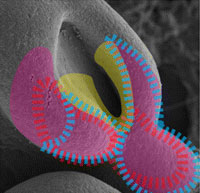
2009. Song L, Li Y, Wang K, Wang YZ, Molotkov A, Gao L, Zhao T, Yamagami T, Wang Y, Gan Q, Pleasure DE, Zhou CJ. Lrp6-mediated canonical Wnt signaling is required for lip formation and fusion. Development 136:3161-3171.
Abstract: Neither the mechanisms that govern lip morphogenesis nor the cause of cleft lip are well understood. We report that genetic inactivation of Lrp6, a co-receptor of the Wnt/beta-catenin signaling pathway, leads to cleft lip with cleft palate. The activity of a Wnt signaling reporter is blocked in the orofacial primordia by Lrp6 deletion in mice. The morphological dynamic that is required for normal lip formation and fusion is disrupted in these mutants. The expression of the homeobox genes Msx1 and Msx2 is dramatically reduced in the mutants, which prevents the outgrowth of orofacial primordia, especially in the fusion site. We further demonstrate that Msx1 and Msx2 (but not their potential regulator Bmp4) are the downstream targets of the Wnt/beta-catenin signaling pathway during lip formation and fusion. By contrast, a ;fusion-resistant' gene, Raldh3 (also known as Aldh1a3), that encodes a retinoic acid-synthesizing enzyme is ectopically expressed in the upper lip primordia of Lrp6-deficient embryos, indicating a region-specific role of the Wnt/beta-catenin signaling pathway in repressing retinoic acid signaling. Thus, the Lrp6-mediated Wnt signaling pathway is required for lip development by orchestrating two distinctively different morphogenetic movements.
 Editorial Highlight: Cleft lip, with or without cleft palate, occurs in around 1 in 700 human newborns, but little is known about the mechanisms involved. Now, on p. 3161, Chengji Zhou and colleagues identify the Wnt co-receptor Lrp6 as being crucial for normal lip morphogenesis in mice. All mice in which Lrp6 is deleted develop a cleft lip with cleft palate. These defects correlate with blocked Wnt/β-catenin signalling and with reduced cell proliferation in the primordia of the lip and palate (known as the orofacial primordia) earlier in development. Concomitantly with reduced Wnt/β-catenin signalling, the expression of the homeobox genes Msx1 and Msx2, which are important for mesenchyme proliferation, is decreased in the orofacial primordia. Conversely, the expression of Raldh3, which encodes a retinoic acid-synthesising enzyme that counteracts tissue fusion, expands. The authors demonstrate that Msx1 and Msx2 are direct targets of Wnt/β-catenin signalling and conclude that Lrp6-mediated Wnt/β-catenin signalling regulates lip formation and fusion by balancing the activities of Msx1 and Msx2 with the opposing activity of Raldh3.
Editorial Highlight: Cleft lip, with or without cleft palate, occurs in around 1 in 700 human newborns, but little is known about the mechanisms involved. Now, on p. 3161, Chengji Zhou and colleagues identify the Wnt co-receptor Lrp6 as being crucial for normal lip morphogenesis in mice. All mice in which Lrp6 is deleted develop a cleft lip with cleft palate. These defects correlate with blocked Wnt/β-catenin signalling and with reduced cell proliferation in the primordia of the lip and palate (known as the orofacial primordia) earlier in development. Concomitantly with reduced Wnt/β-catenin signalling, the expression of the homeobox genes Msx1 and Msx2, which are important for mesenchyme proliferation, is decreased in the orofacial primordia. Conversely, the expression of Raldh3, which encodes a retinoic acid-synthesising enzyme that counteracts tissue fusion, expands. The authors demonstrate that Msx1 and Msx2 are direct targets of Wnt/β-catenin signalling and conclude that Lrp6-mediated Wnt/β-catenin signalling regulates lip formation and fusion by balancing the activities of Msx1 and Msx2 with the opposing activity of Raldh3.
2009. Yamagami T, Molotkov A, Zhou CJ. Canonical Wnt signaling activity during synovial joint development. Journal of Molecular Histology 40:311-316.
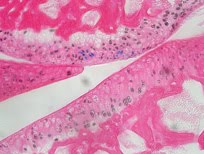 Abstract: Wnt signaling plays important roles in skeletal development. However, the activation and function of canonical Wnt signaling in joint development remains unclear. We analyzed the lineage identity and developmental changes of the Wnt-responsive cells during synovial joint formation as well as adulthood in the Wnt signaling reporter TOPgal transgenic mice. At embryonic day (E) 12.5, we found that the TOPgal was inactivated in the presumptive joint forming interzone, but it was intensively activated in the cartilage anlage of developing long bones and digits. At E14.5, the TOPgal activity was found in a subgroup of the articular chondrocyte lineage cells, which were co-immunolabeled with Doublecortin intensively and with Vinculin weakly. At E18.5, the TOPgal/Doublecortin co-immunolabeled cells were found in the superficial layer of the developing articular cartilage. During postnatal development, the TOPgal(+) articular chondrocytes were abundant at P7 and decreased from P10. A small number of TOPgal(+) articular chondrocytes were also found in adult joints. Our study suggests an age- and lineage-specific role of canonical Wnt signaling in joint development and maintenance.
Abstract: Wnt signaling plays important roles in skeletal development. However, the activation and function of canonical Wnt signaling in joint development remains unclear. We analyzed the lineage identity and developmental changes of the Wnt-responsive cells during synovial joint formation as well as adulthood in the Wnt signaling reporter TOPgal transgenic mice. At embryonic day (E) 12.5, we found that the TOPgal was inactivated in the presumptive joint forming interzone, but it was intensively activated in the cartilage anlage of developing long bones and digits. At E14.5, the TOPgal activity was found in a subgroup of the articular chondrocyte lineage cells, which were co-immunolabeled with Doublecortin intensively and with Vinculin weakly. At E18.5, the TOPgal/Doublecortin co-immunolabeled cells were found in the superficial layer of the developing articular cartilage. During postnatal development, the TOPgal(+) articular chondrocytes were abundant at P7 and decreased from P10. A small number of TOPgal(+) articular chondrocytes were also found in adult joints. Our study suggests an age- and lineage-specific role of canonical Wnt signaling in joint development and maintenance.
2008. Zhou CJ, Molotkov A, Song L, Li Y, Pleasure DE, Pleasure SJ, Wang YZ. Ocular coloboma and dorsoventral neuroretinal patterning defects in Lrp6 mutant eyes. Developmental Dynamics 237:3681-3689.
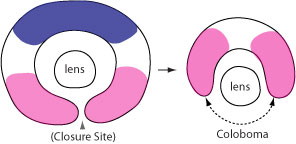 Abstract: Coloboma, an ocular birth defect seen in humans and other species, is caused by incomplete closure of the optic fissure. Here, we demonstrate that genetic deletion of Lrp6, a bottleneck coreceptor in the canonical Wnt signaling pathway, results in ocular coloboma and neuroretinal patterning defects in mice. The expression of ventral neuroretinal patterning gene Vax2 was conserved but with dorsally shifted expression domains; however, the dorsal neuroretinal patterning gene Tbx5 was lost in the Lrp6-mutant eyes at embryonic day 10.5. Both Bmp4 and phosphorylated Smad 1/5/8 were also significantly attenuated in the dorsal neuroretina. In addition, the retinoic acid synthesizing enzymes Raldh1 and Raldh3 were significantly changed in the mutant eyes. Our findings suggest that defective retinal patterning causes coloboma in the Lrp6-deficient mice, and that canonical Wnt signaling plays a primary role in dorsal neuroretinal patterning and related morphogenetic movements by regulation of both Bmp and retinoic acid signaling pathways.
Abstract: Coloboma, an ocular birth defect seen in humans and other species, is caused by incomplete closure of the optic fissure. Here, we demonstrate that genetic deletion of Lrp6, a bottleneck coreceptor in the canonical Wnt signaling pathway, results in ocular coloboma and neuroretinal patterning defects in mice. The expression of ventral neuroretinal patterning gene Vax2 was conserved but with dorsally shifted expression domains; however, the dorsal neuroretinal patterning gene Tbx5 was lost in the Lrp6-mutant eyes at embryonic day 10.5. Both Bmp4 and phosphorylated Smad 1/5/8 were also significantly attenuated in the dorsal neuroretina. In addition, the retinoic acid synthesizing enzymes Raldh1 and Raldh3 were significantly changed in the mutant eyes. Our findings suggest that defective retinal patterning causes coloboma in the Lrp6-deficient mice, and that canonical Wnt signaling plays a primary role in dorsal neuroretinal patterning and related morphogenetic movements by regulation of both Bmp and retinoic acid signaling pathways.
2008. Wang YZ, Molotkov A, Song L, Li Y, Pleasure DE, Zhou CJ. Activation of the Wnt/beta-catenin signaling reporter in developing mouse olfactory nerve layer marks a specialized subgroup of olfactory ensheathing cells. Developmental Dynamics 237:3157-3168.
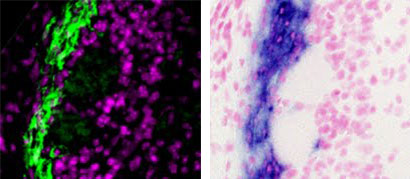
Abstract: Wnt reporter TOPgal mice carry a beta-galactosidase (betagal) gene under the control of the Wnt/beta-catenin signaling responsive elements. We found that the intensely immunolabeled betagal+ cells were co-immunolabeled with Nestin and formed a tangentially oriented single-cell layer in the "connecting or docking zone" where the olfactory sensory axons attached to the brain surface during mid-gestation. During early postnatal development, betagal+ cells were located in the inner olfactory nerve layer (ONLi) and co-labeled with olfactory ensheathing cell (OEC) markers S100beta and NPY but not with lineage-specific markers for neurons, oligodendrocytes, astrocytes, and microglia, demonstrating that the TOPgal marked a subpopulation of OECs. By confocal microscopy, we found that TOPgal activated processes extended along the developing glomerulus and formed multiple tunnel-like structures that ensheathe and bridge olfactory sensory axonal bundles from ONLi to the glomerulus, which may play a key role in glomerulus formation and convergent sorting of the peripheral olfactory axons.