Areas of Research
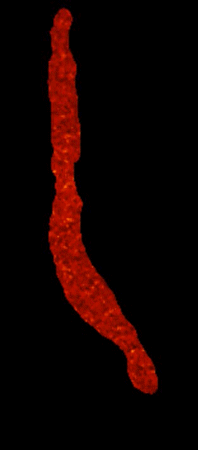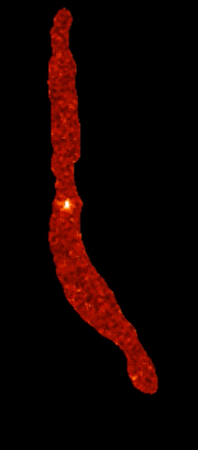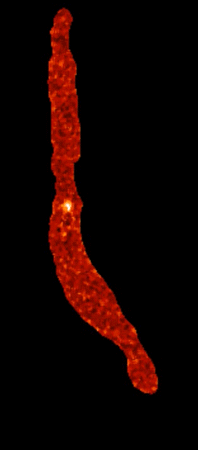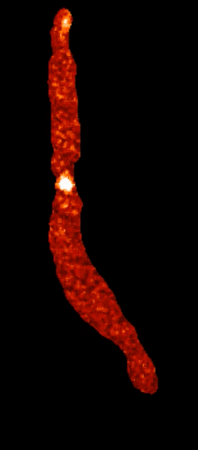
Action potential-evoked calcium transients in a ventricular myocyte |
The heart continuously pumps blood to ensure that oxygen and nutrients reach the brain, organs, and peripheral tissue. This occurs because action potentials that originate in pace-making cells in the sinoatrial node propagate via gap junctions through the atria and ventricles of the heart to evoke contraction in a precise sequence. This chain of events is known as excitation-contraction (EC) coupling. Related events that modulate cardiac gene expression proceed via excitation-transcription (ET) coupling pathways. Both processes can contribute to arrhythmogenic disorders, a major cause of sudden death in otherwise healthy individuals. The work of the heart would be futile if it was not for arteries, which deliver the blood pumped by the heart to each organ in our body. Ultimately, the contractile state of the smooth muscle lining the walls of arteries determine arterial diameter and hence blood flow. Like cardiac myocytes, arterial myocyte contraction is controlled by changes in membrane potential and Ca2+ influx. Our broad goal is to understand the biophysical and molecular mechanisms the regulate ET and EC coupling in cardiac and arterial smooth muscle during physiological conditions and during the development of hypertension and heart failure. This is important because cardiovascular disease is the major cause of death in the United States and other developed countries We use a multidisciplinary approach in our research, which includes patch-clamp electrophysiology, optogenetics, super resolution, confocal and TIRF imaging as well as telemetry and molecular biological techniques. |
Calcium sparklets in an arterial myocyte |